Host-Microbe Interactions
Mission
The Teixeira laboratory studies the interactions between hosts and microbes. These range from antagonistic, where the microbes are pathogenic, to beneficial ones. The group aims to understand how the relationships are specified and regulated, as well as, how the partners influence each other in terms of physiology, ecology, and evolution.
Importance
Most animals live in association with microbes, including fungi, bacteria, and viruses. Humans, for instance, carry a trillion (1012) bacteria in their guts under normal, healthy conditions. Many of these microbes are beneficial to the host, influencing from nutrition to resistance to pathogens. Therefore, these interactions impact the physiology, the ecology, and the evolution of microbes and hosts. Using Drosophila melanogaster as a model system we want to understand fundamental principles of these interactions across different organisms, including humans. Some discoveries can be applied to improve human health, as, for example, the use of the bacteria Wolbachia in mosquitoes to prevent transmission of dengue and other arboviruses.
Main research interests
One focus of the Host-Microbe Interactions laboratory is how D. melanogaster resists viral infections. We showed that Toll, one of the main immune pathways, is important to resist viral oral infections. This is one of strongest, general, antiviral pathways in Drosophila. The research focuses now on how is Toll activated by viral infection, what are its antiviral effector mechanisms, and also in new antiviral pathways.
We also found the intracellular endosymbiotic bacteria Wolbachia provides strong resistance to viruses. Wolbachia is the most common intracellular bacteria in insects, and probably animals. It is transmitted maternally and can induce a wide range of phenotypes to its hosts, being protection to viruses one of them. This protection has a practical application since Wolbachia introduction to the mosquito Aedes aegypti leads to resistance to dengue, and other arboviruses. The release of these Wolbachia-carrying mosquitoes has been successful in diminishing dengue transmission in endemic cities, and is being deployed worldwide.

Gut bacteria form clusters in the anterior part of the gut of Drosophila. Green - Acetobacter thailandicus, cyan - DNA. Image from Pais et al. PLOS Biology 2018.
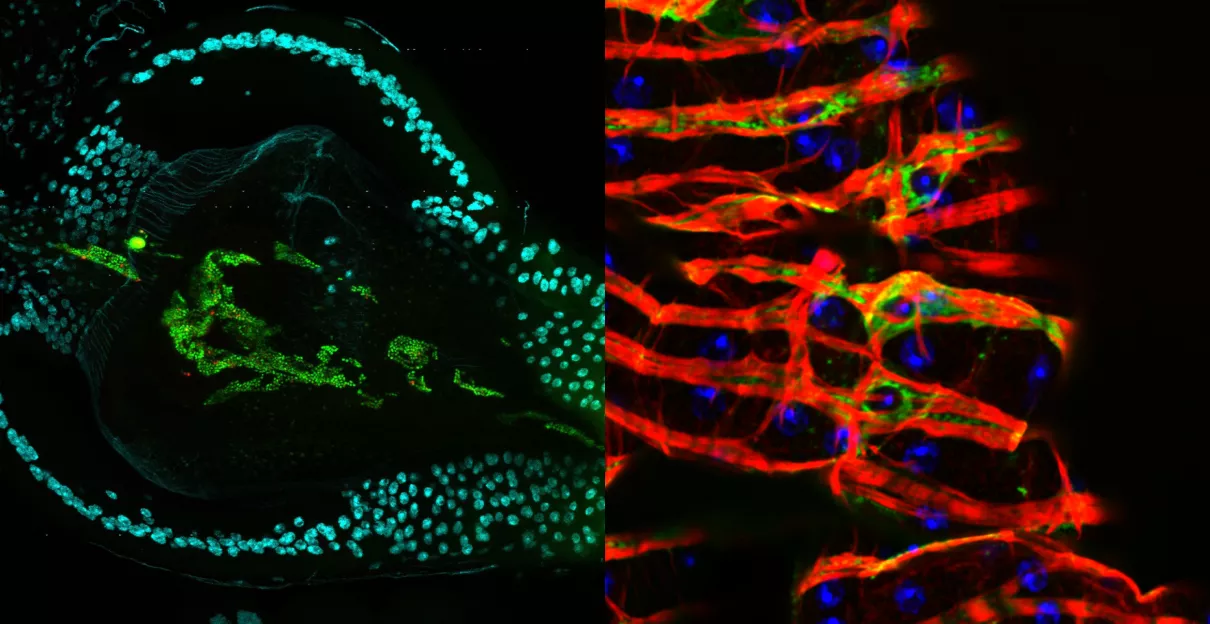
Viral infection in the visceral muscle of Drosophila. Green - Drosophila C virus, red - Actin , blue - DNA.
We are interested in understanding what is the mechanism of antiviral protection and which factors influence it (e.g. temperature, Wolbachia titres). Moreover, we want to know how the growth of Wolbachia is controlled from the bacteria and host perspective. This is important not only from fundamental point of view but also on the design of long-term successful strategies of Wolbachia-carrying mosquitoes deployment.
Finally, we are interested in studying the interaction of Drosophila with gut bacteria. We are studying how these strongly impact the growth of larvae and fertility of adults. Moreover, this system allows us to investigate which factors regulate the specificity and capacity of bacteria to colonize the gut of animals.
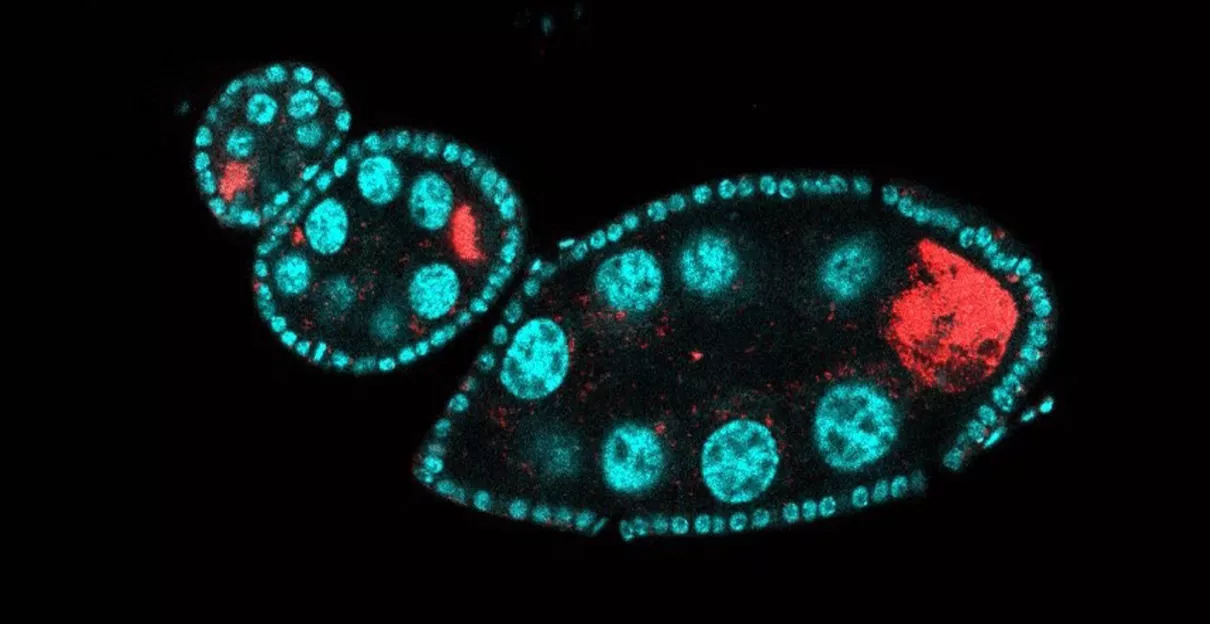
Wolbachia localizes to the oocyte in order to be transmitted to the next generation. Red - Wolbachia, cyan - DNA.
Tools and models
The laboratory uses Drosophila melanogaster as a model system because of its powerful genetics, extensive knowledge on its biology, relatively simple microbiota composition, and capacity of producing axenic and gnotobiotic flies (without any, or with specified associated microbes).
The laboratory mainly works with microbes that are known to naturally interact with D. melanogaster. These are more prone to be complex and co-evolved interactions. The microbes used include viruses (e.g. Drosophila C virus), gut bacteria (e.g. Acetobacter spp.), and the endosymbiont Wolbachia.
These interactions are analysed from multiple approaches: biochemistry, cell biology, genetics, genomics, metabolomics. The laboratory aims to understand the interactions from a molecular, functional perspective in laboratory experiments, but also their ecology and evolution in natural populations.
Funding
- ERC consolidator Grant WOLBAKIAN- Functional genetics of Wolbachia proliferation and protection to viruses
- SymbNET - Genomics and Metabolomics in a Host-Microbe Symbiosis Network Twinning grant from European Union’s Horizon 2020 research and innovation programme
- Advanced Training Course in Host-Microbe Symbiosis Grant from Moore Foundation